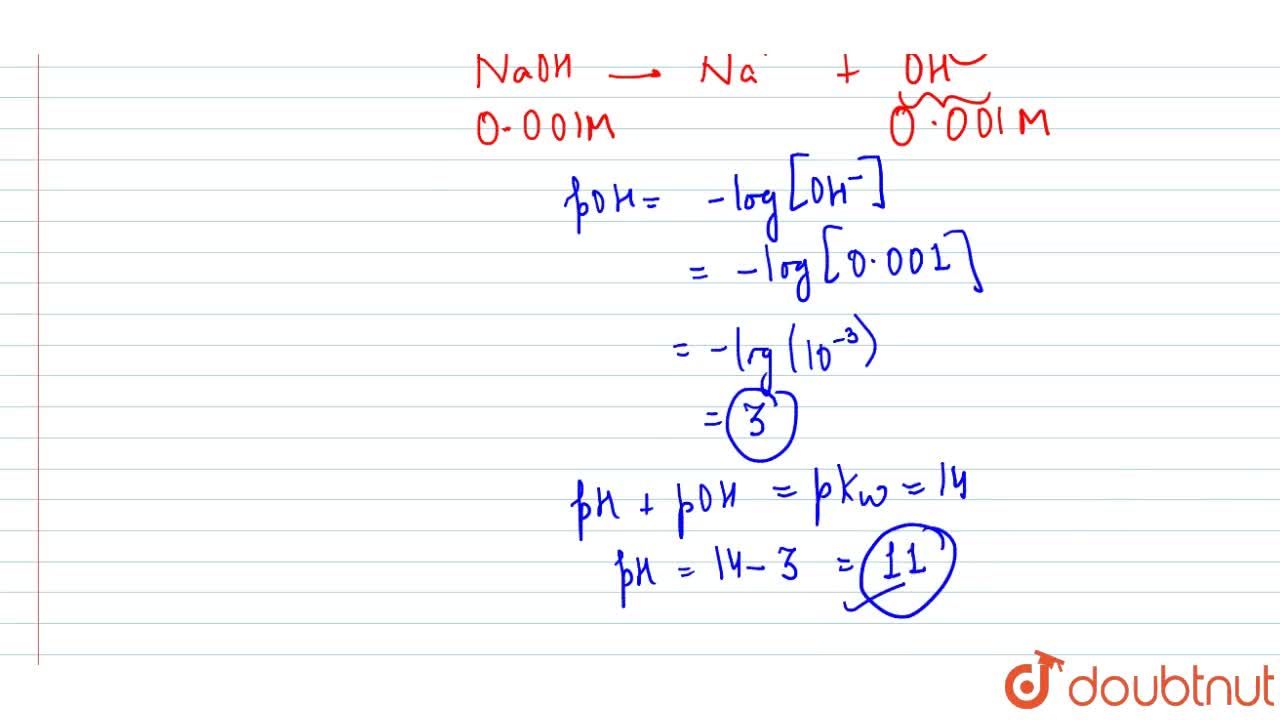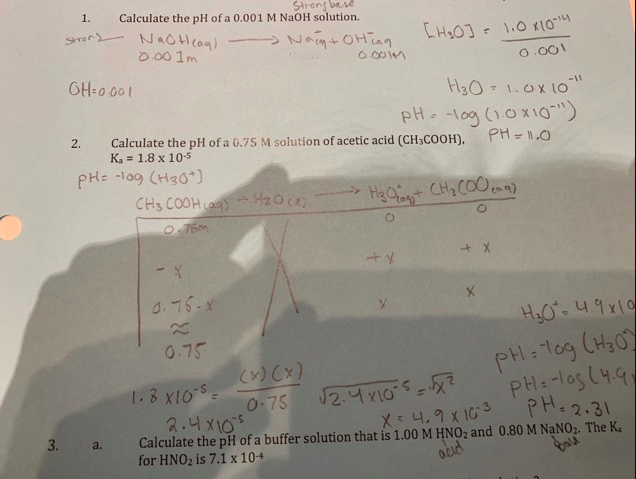Assuming complete dissociation, calculate the pH of the following solutions:(a) 0.003 M HCl (b) 0.005 M NaOH (c) 0.002 M HBr (d) 0.002 M KOH
![SOLVED: Q: What are the [H+], [OH-], pH and pOH of a 0.002M solution of HNO3? Nitric acid v strong acid Completely ionizes in water HNO3 7 Ht + NO3 pH + SOLVED: Q: What are the [H+], [OH-], pH and pOH of a 0.002M solution of HNO3? Nitric acid v strong acid Completely ionizes in water HNO3 7 Ht + NO3 pH +](https://cdn.numerade.com/ask_images/096d2e96c5f04ffb9944004e8f1d5fba.jpg)
SOLVED: Q: What are the [H+], [OH-], pH and pOH of a 0.002M solution of HNO3? Nitric acid v strong acid Completely ionizes in water HNO3 7 Ht + NO3 pH +
What is the pH of the resulting solution when the equal volume of 0.1M Noah and 0.01M HCL are mixed? - Quora
Calculate pH for : (a) 0.001 N NaOH, (b) 0.01 N Ca(OH)2 - Sarthaks eConnect | Largest Online Education Community
Calculate pH for : (a) 0.001 N NaOH, (b) 0.01 N Ca(OH)2 - Sarthaks eConnect | Largest Online Education Community
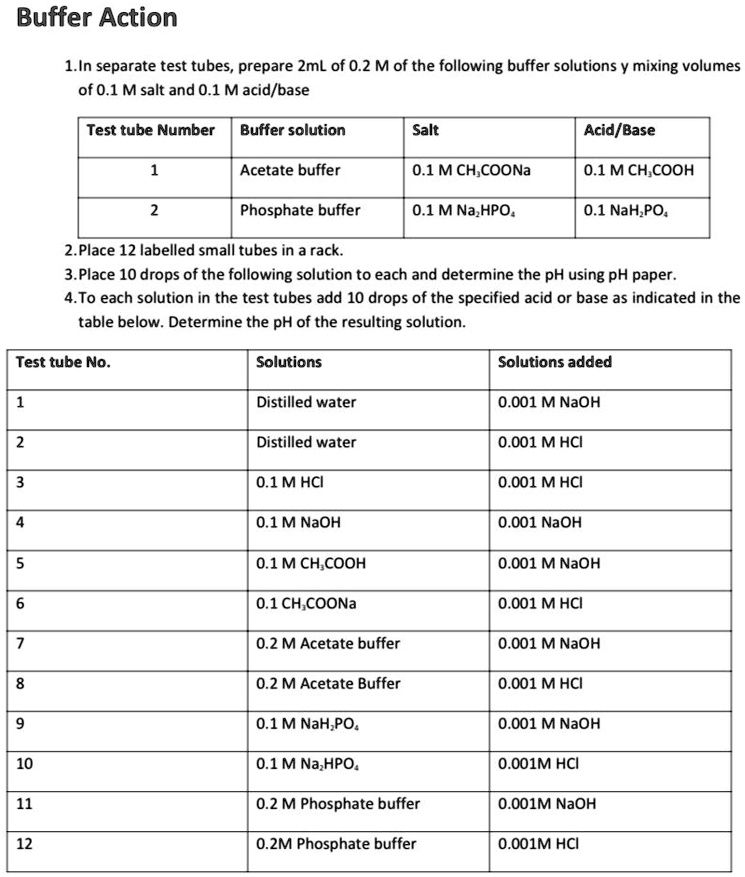
SOLVED: Buffer Action 1.In separate test tubes, prepare ZmL of 0.2 M of the following buffer solutions mixing volumes of 0.1 M salt and 0.1 Macid/base Test tube Number Buffer solution Salt
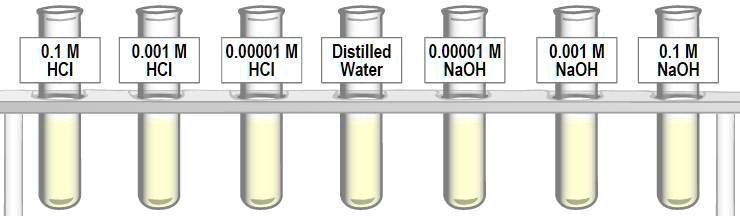
SOLVED: 'What are the pH of these solutions? 0.1 M HCI 0.001 M HCI 0.00001 M HCI Distilled Water 0.00001 M NaOH 0.001 M NaOH 0.1 M NaOH'

Calculate pH for: (a) 0.001 NaOH, (b) 0.01N Ca(OH)(2), (c ) 0.01M Ca(OH)(2), (d) 10^(-8)M NaOH, (e ) 10^(2)MNaOH, (f) 0.0008MMg(OH)(2) Assume complete ionisation of each.